 |
 |
| 1. |
Bioinformatics |
 |
Bioinformatics is a comprehensive research field that deals with a wide range of biological phenomena from the protein, cellular, and individual levels based on gene sequence information using computational information processing. In our laboratory, we are working on the creation of tailor-made medicine by using gene expression analysis methods such as next-generation sequencers. Our research has made it possible to analyze gene expression in patients using DNA microarrays and other methods, and to formulate appropriate treatment plans for diseases. We will continue to promote our research from various angles. |
| 2. |
Structural Life Science |
 |
Structural life science is a research field that aims to elucidate biological phenomena interwoven by the correlation between the structure and function of biological macromolecules (nucleic acids, proteins, carbohydrates, and lipids) using structural analysis techniques (X-ray diffraction, electron microscopy, NMR, etc.) and computational chemistry. Our laboratory uses computer simulations (molecular dynamics, electrostatic complementarity, shape complementarity, and quantum chemical calculations) to approach the elucidation of biological phenomena. So far, we have clarified the transcriptional activation mechanism of Notch signaling, which plays an important role in cell fate determination, and the cell cycle regulation mechanism of fungi. In the future, we will continue to elucidate biological phenomena one by one to answer the question "What is life? We will continue to elucidate biological phenomena one by one to answer the question, "What is life?"
|
| 3. |
Creation of Molecular Targeted Drugs |
 |
Conventional drug development has been carried out by relying on the intuition and experience of a limited number of specialists. It is said that it takes an average of 15 years and costs about 50 billion yen to develop a single drug. In our laboratory, we are promoting computer-aided medical drug discovery, so-called IT drug discovery, which aims to create drugs logically and efficiently. |
 |
 |
 |
A. Antibody Drugs |
 |
| | Based on gene sequence and structural information, we have developed an antibody drug (fusion of the extracellular region of IL-10R1 and the invariant region of the antibody) for the treatment of malignant melanoma. | |
 |
B. Peptide Drugs |
 |
| | We have developed an "in silico molecular evolution method" that executes the "phage display method" (in vitro molecular evolution method), which is a genetic engineering method, on a computer to create peptide drugs useful for the treatment of spinal cord injuries. | |
 |
C. Small molecule drugs |
 |
| | By utilizing high-throughput screening using a super computing system, we have searched for functional control molecules that are promising as therapeutic agents for childhood cancer.
We are currently investigating the potential of approximately 500 compounds (as of October 2014) registered in the "Chiba University Original Compound Library" as potential molecular targeted therapies. We will continue to make full use of computer simulations (molecular dynamics calculations, electrostatic complementarity calculations, shape complementarity calculations, and quantum chemical calculations) to create new molecular targeted drugs.
| |
| 4. |
Creation of non-invasive medicine by photoimmunotherapy |
 |
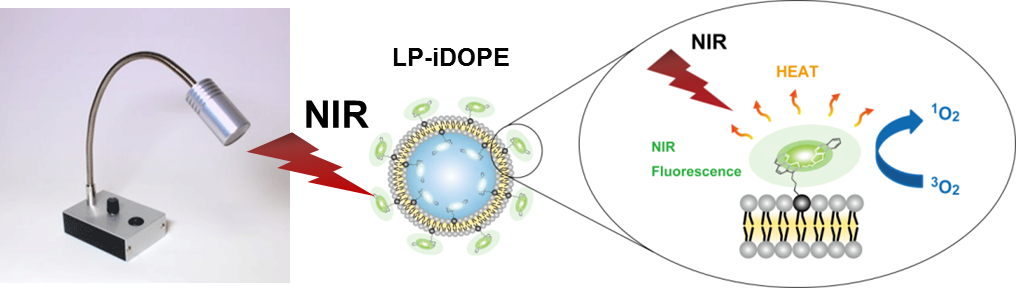
In order to realize safer and more secure medical technology, we are developing combination products that combine nanodevices (pharmaceuticals) and optical devices (medical devices), and promoting efforts to develop therapies that contribute to improving patients' QOL. We have already developed a liposome formulation (LP-iDOPE) that contains indocyanine green (ICG), a photosensitizer, as a nanodevice, and confirmed specific accumulation in tumor tissues by the EPR effect (leakage of 20-200 nm particles from immature vascular tissues around cancer tissues). In addition, we have developed an LED therapy device that generates near-infrared light as an optical device. In the future, we will promote translational research for non-invasive treatment by photoimmunotherapy using the optical absorption property of ICG. |
|
|
|
 |
|
 |
| Copyright(C) 2009- Dept. of Bioinfor., Chiba Univ. All Rights Reserved. |
|
|
|
